A carbon atom can form four single covalent bonds simultaneously. 
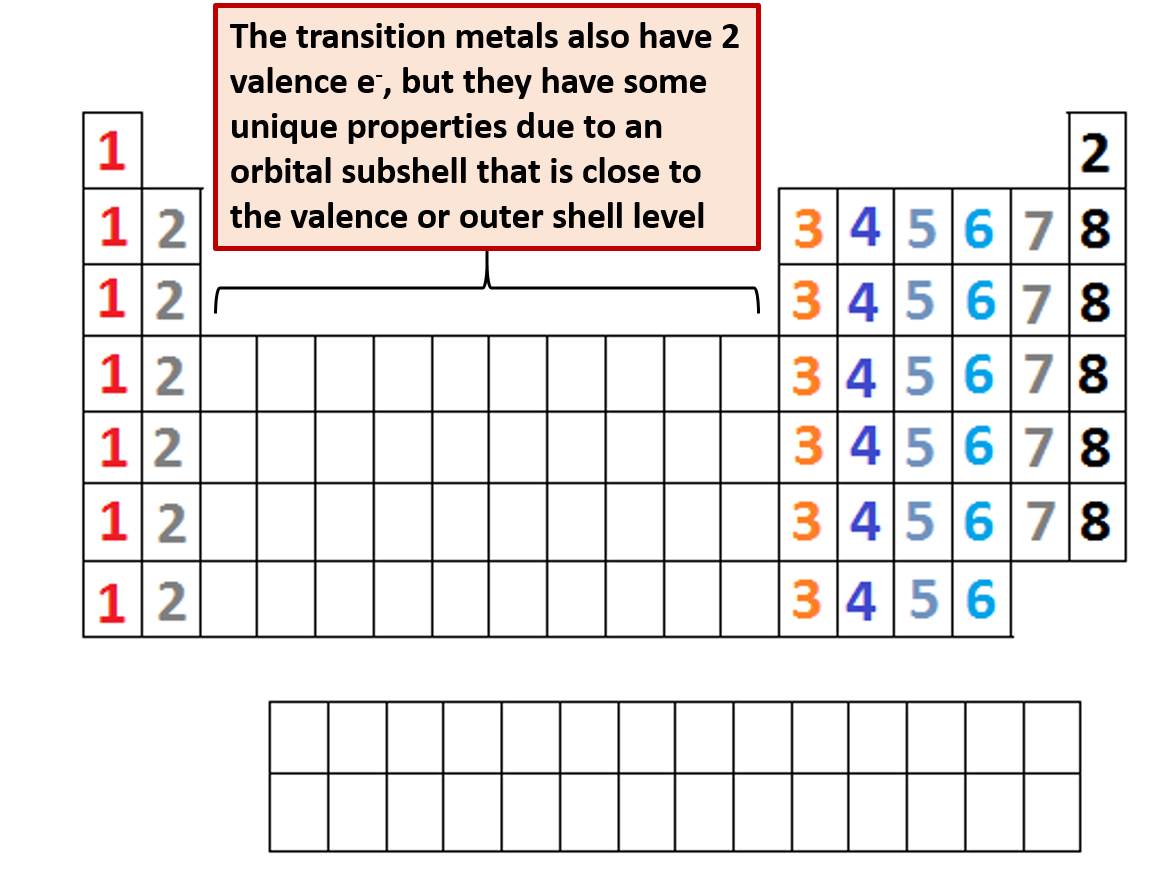
A carbon atom cannot form two double bonds simultaneously. Carbon can form a triple covalent bond with another carbon atom. Saturated Unsaturated and SupersaturatedĪpproximately 3.4 billion years ago, the first life form sprung up in a hydrothermal vent deep in the ocean near present-day Quebec. To form ionic bonds, Carbon molecules must either gain or lose 4 electrons &This is highly unfavorable. If a carbon atom has four valence electrons, which of the following statements are true I.Reaction Quotient and Le Chatelier's Principle.Prediction of Element Properties Based on Periodic Trends.Molecular Structures of Acids and Bases.Ion and Atom Photoelectron Spectroscopy.Elemental Composition of Pure Substances.Application of Le Chatelier's Principle.Structure, Composition & Properties of Metals and Alloys.Intramolecular Force and Potential Energy Valence electrons are the electrons in the highest occupied principal energy level of an atom.Variable Oxidation State of Transition Elements.Transition Metal Ions in Aqueous Solution.The atomic number of each element increases by one, reading from left to right. Period A horizontal row in the periodic table. Members of a group typically have similar properties and electron configurations in their outer shell. Table 2.6. 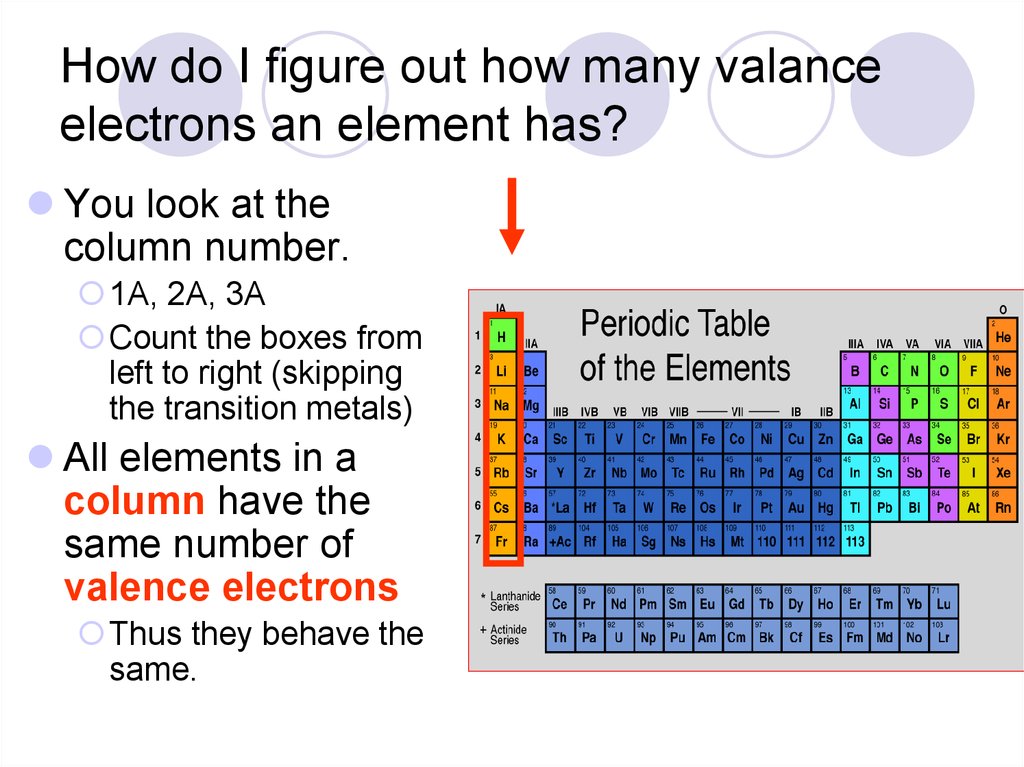
The number of valence electrons for each main group element can be determined by the column, or group, it occupies on the periodic table. Single and Double Replacement Reactions 6 12.011 Glossary Group A vertical column in the periodic table. Carbon has 6 electrons - 2 in the first shell, and 4 in the second shell (so four valence electrons). 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
